What Term Is Used To Describe Genetically Altered Animals?
GloFish are the first transgenic animals bachelor to the American public. But what's the biotechnology behind them?

Effigy 1: The multicolored GloFish®.
Courtesy of www.glofish.com. All rights reserved. ![]()
"Seeing is believing with GloFish. They are absolutely stunning!" The preceding is some of the marketing textile you'd read if you visited the GloFish website (GloFish, 2008). Dazzler may exist in the eye of the beholder, but almost anybody would agree that these get-go—and, and then far, only—transgenic animals made available to the general public in the Us (except in California, pending a formal review of their potential effect on the environment) are a worthy conversation piece. A transgenic, or genetically modified, organism is one that has been altered through recombinant Dna technology, which involves either the combining of Dna from different genomes or the insertion of foreign DNA into a genome. GloFish (Figure 1) are a blazon of transgenic zebrafish ( Danio rerio ) that have been modified through the insertion of a green fluorescent protein (gfp) gene. Non all GloFish are green, withal. Rather, there are several gfp gene constructs, each encoding a different colored phenotype, from fluorescent yellow to fluorescent cherry-red.
Currently, GloFish are the only recombinant-DNA animal that has been approved for homo "employ" by the U.S. Food and Drug Assistants. Their approval has raised of import questions almost whether, and to what extent, genetically modified animals should be made available to consumers. Only how were scientists able to create these engineered organisms in the showtime place? Like so many genetic technologies used today, recombinant DNA technology had its origins in the late 1960s and early on 1970s. By the 1960s, scientists had already learned that cells repair Dna breaks by reuniting, or recombining, the broken pieces. Thus, it was just a matter of time before researchers identified the raw biological ingredients necessary for recombination, figured out how those ingredients functioned together, and so tried to govern the recombining procedure themselves.
Early on Experiments Provide the Footing for Recombinant Organisms
Although recombinant Dna technology first emerged in the 1960s and 1970s, the basic principle of recombination had been discovered many years before. Indeed, in 1928, Frederick Griffith, an English language medical officer studying the bacteria responsible for a pneumonia epidemic in London, outset demonstrated what he termed "genetic transformation"; here, living cells took up genetic material released by other cells and became phenotypically "transformed" past the new genetic information. More than a decade later, Oswald Avery repeated Griffith's work and isolated the transforming molecule, which turned out to be Deoxyribonucleic acid. These experiments showed that DNA can be transferred from one jail cell to another in the laboratory, thus changing the actual genetic phenotype of an organism.
Prior to these archetype experiments, the thought that the genetic material was a specific chemical that could be modified and transferred into cells was certainly controversial. But before the explosion in recombinant DNA could brainstorm, scientists would have to learn not only how to transfer DNA, just also how to isolate and alter individual genes.
Key Developments in Recombinant DNA Technology
Following these early experiments, 4 key developments helped lead to construction of the first recombinant Dna organism (Kiermer, 2007). The starting time two developments revolved around how scientists learned to cut and paste pieces of DNA from unlike genomes using enzymes. The latter 2 events involved the evolution of techniques used to transfer foreign DNA into new host cells.
Discovering the Cutting-and-Paste Enzymes
The first major pace forwards in the ability to chemically modify genes occurred when American biologist Martin Gellert and his colleagues from the National Institutes of Health purified and characterized an enzyme in Escherichia coli responsible for the actual joining, or recombining, of separate pieces of Deoxyribonucleic acid (Zimmerman et al., 1967). They called their find "DNA-joining enzyme," and this enzyme is at present known every bit DNA ligase. All living cells utilise some version of Deoxyribonucleic acid ligase to "glue together" short strands of DNA during replication. Using E. coli extract, the researchers next showed that merely in the presence of ligase was it possible to repair unmarried-stranded breaks in λ phage Dna. (Discovered in 1950 by American microbiologist Esther Lederberg, λ phage is a virus particle that infects East. coli.) More specifically, they showed that the enzyme was able to course a 3'-5'-phosphodiester bail between the 5'-phosphate cease of the last nucleotide on one DNA fragment and the three'-OH end of the last nucleotide on an adjacent fragment. The identification of Deoxyribonucleic acid ligase was the first of several key steps that would somewhen empower scientists to endeavor their ain recombination experiments—experiments that involved non just recombining the DNA of a single individual, but recombining Dna from dissimilar individuals, including different species.
A second major step forrad in gene modification was the discovery of restriction enzymes, which cleave Dna at specific sequences. These enzymes were discovered at approximately the same time as the first Dna ligases by Swiss biologist Werner Arber and his colleagues while they were investigating a phenomenon called host-controlled brake of bacteriophages. Bacteriophages are viruses that invade and oftentimes destroy their bacterial host cells; host-controlled restriction refers to the defense mechanisms that bacterial cells have evolved to bargain with these invading viruses. Arber's team discovered that one such mechanism is enzymatic activeness provided past the host cell. The team named the responsible enzymes "restriction enzymes" considering of the fashion they restrict the growth of bacteriophages. These scientists were likewise the outset to demonstrate that brake enzymes damage invading bacteriophages past cleaving the phage Deoxyribonucleic acid at very specific nucleotide sequences (at present known as restriction sites). The identification and label of restriction enzymes gave biologists the ways to cut specific pieces of Deoxyribonucleic acid required (or desired) for subsequent recombination.
Inserting Strange Deoxyribonucleic acid into a New Host Jail cell
Although Griffith and Avery had had demonstrated the power to transfer foreign genetic material into cells decades before, this "transformation" was very inefficient, and it involved "natural" rather than manipulated DNA. Only in the 1970s did scientists begin to use vectors to efficiently transfer genes into bacterial cells. The commencement such vectors were plasmids, or modest Dna molecules that alive naturally inside bacterial cells and replicate separately from a bacterium's chromosomal Deoxyribonucleic acid.
Plasmids' utility as a DNA shuttle, or vector, was discovered by Stanford University biochemist Stanley Cohen. Scientists had already established that some bacteria had what were known as antibody resistance factors, or R factor-plasmids that replicated independently within the bacterial cell. Merely scientists knew little well-nigh how the different R factor genes functioned. Cohen thought that if at that place were an experimental system for transforming host bacterial cells with these R-gene DNA molecules, he and other researchers might be able to better empathise R-factor biological science and figure out exactly what it was most these plasmids that made bacteria antibiotic-resistant. He and his colleagues developed that system by demonstrating that calcium chloride-treated E. coli can be genetically transformed into antibiotic-resistant cells by the improver of purified plasmid DNA (in this example, purified R-factor DNA) to the bacteria during transformation (Cohen et al., 1972).
Recombinant Plasmids in Leaner
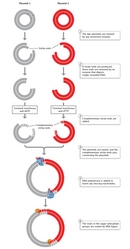
The following twelvemonth, Stanley Cohen and his colleagues were as well the starting time to construct a novel plasmid Deoxyribonucleic acid from ii separate plasmid species which, when introduced into Due east. coli, possessed all the nucleotide base of operations sequences and functions of both parent plasmids. Cohen'south team used restriction endonuclease enzymes to cleave the double-stranded DNA molecules of the two parent plasmids. The team next used Dna ligase to rejoin, or recombine, the DNA fragments from the 2 different plasmids (Effigy 2). Finally, they introduced the newly recombined plasmid Deoxyribonucleic acid into E. coli. The researchers were able to bring together ii DNA fragments from completely different plasmids because, as they explained, "the nucleotide sequences cleaved are unique and self-complementary so that Deoxyribonucleic acid fragments produced past one of these enzymes can associate by hydrogen-bonding with other fragments produced by the aforementioned enzyme" (Cohen et al., 1973).

The aforementioned could be said of any Deoxyribonucleic acid—not just plasmids—from two different species. This universality—the capacity to mix and lucifer Dna from different species, because Dna has the aforementioned construction and function in all species and considering brake and ligase enzymes cutting and paste the same ways in unlike genomes—makes recombinant DNA biology possible.
Today, the E. coli λ bacteriophage is one of the most widely used vectors used to carry recombinant Dna into bacterial cells. This virus makes an excellent vector considering almost one-third of its genome is considered nonessential, meaning that it can exist removed and replaced by foreign Deoxyribonucleic acid (i.eastward., the Dna being inserted). As illustrated in Figure 3, the nonessential genes are removed past restriction enzymes (the specific restriction enzyme EcoRI is shown in the effigy), the foreign DNA inserted in their place, and so the last recombinant Deoxyribonucleic acid molecule is packaged into the virus's protein coat and prepped for introduction into its host cell.
Vectors Used in Mammalian Cells
A quaternary major step forward in the field of recombinant Dna technology was the discovery of a vector for efficiently introducing genes into mammalian cells. Specifically, researchers learned that recombinant DNA could be introduced into the SV40 virus, a pathogen that infects both monkeys and humans. Indeed, in 1972, Stanford Academy researcher Paul Berg and his colleagues integrated segments of λ phage DNA, besides as a segment of E. coli DNA containing the galactose operon, into the SV40 genome. (The E. coli galactose operon is a cluster of genes that plays a role in galactose sugar metabolism.) The significance of their achievement was its demonstration that recombinant Dna technologies could be applied to essentially any DNA sequences, no matter how distantly related their species of origin. In their words, these researchers "developed biochemical techniques that are generally applicative for joining covalently any ii DNA molecules" (Jackson et al., 1972). While the scientists didn't actually introduce foreign Deoxyribonucleic acid into a mammalian cell in this experiment, they provided (proved) the means to practise and then.
Recombinant Dna Technology Creates Recombinant Animals
The first actual recombinant animal cells weren't developed until about a decade after the research conducted by Berg's team, and most of the early studies involved mouse cells. In 1981, for example, Franklin Costantini and Elizabeth Lacy of the University of Oxford introduced rabbit DNA fragments containing the developed beta globin gene into murine (mouse) germ-line cells (Costantini & Lacy, 1981). (The beta globins are a family of polypeptides that serve equally the subunits of hemoglobin molecules.) Some other group of scientists had demonstrated that foreign genes could exist successfully integrated into murine somatic cells, but this was the starting time demonstration of their integration into germ cells. In other words, Costantini and Lacy were the first to engineer an entire recombinant animal (albeit with relatively low efficiency).
Interestingly, non long afterwards the publication of his team's 1972 study, Paul Berg led a voluntary moratorium in the scientific customs against certain types of recombinant Deoxyribonucleic acid inquiry. Clearly, scientists take always been aware that the ability to manipulate the genome and mix and match genes from different organisms, even different species, raises immediate and serious questions nearly the potential hazards and risks of doing then—implications still being debated today.
Since these early studies, scientists take used recombinant Deoxyribonucleic acid technologies to create many different types of recombinant animals, both for scientific study and for the profitable manufacturing of man proteins. For instance, mice, goats, and cows accept all been engineered to create medically valuable proteins in their milk; moreover, hormones that were once isolated only in small-scale amounts from man cadavers can now be mass-produced past genetically engineered cells. In fact, the entire biotechnology industry is based upon the power to add new genes to cells, plants, and animals As scientists notice important new proteins and genes, these technologies will continue to form the foundation of future generations of discoveries and medical advances.
References and Recommended Reading
Cohen, S. N., et al. Nonchromosomal antibiotic resistance in bacteria: Genetic transformation of Escherichia coli by R-cistron Dna. Proceedings of the National University of Sciences 69, 2110–2114 (1972)
———. Structure of biologically functional bacterial plasmids in vitro. Proceedings of the National University of Sciences seventy, 3240–3244 (1973)
Costantini, F., & Lacy, East. Introduction of a rabbit beta-globin gene into the mouse germ line. Nature 294, 92–94 (1981) (link to commodity)
Crea, R., et al. Chemical synthesis of genes for homo insulin. Proceedings of the National Academy of Sciences 75, 5765–5769 (1978)
GloFish. GloFish habitation page. www.glofish.com (Accessed July three, 2008)
Jackson, D. A., et al. Biochemical method for inserting new genetic information into Deoxyribonucleic acid of simian virus 40: Circular SV40 DNA molecules containing lambda phage genes and the galactose operon of Escherichia coli. Proceedings of the National Academy of Sciences 69, 2904–2909 (1972)
Kiermer, V. The dawn of recombinant DNA. Nature Milestones: DNA Technologies, http://www.nature.com/milestones/miledna/full/miledna02.html (2007) (link to article)
Miller, H. I. FDA on transgenic animals—A canis familiaris's breakfast? Nature Biotechnology 26, 159–160 (2008) (link to commodity)
Zimmerman, S. B., et al. Enzymatic joining of DNA strands: A novel reaction of diphosphopyridine nucleotide. Proceedings of the National Academy of Sciences 57, 1841–1848 (1967)
Source: http://www.nature.com/scitable/topicpage/recombinant-dna-technology-and-transgenic-animals-34513
Posted by: deckertoomeng.blogspot.com

0 Response to "What Term Is Used To Describe Genetically Altered Animals?"
Post a Comment